What we achieved
The Solution
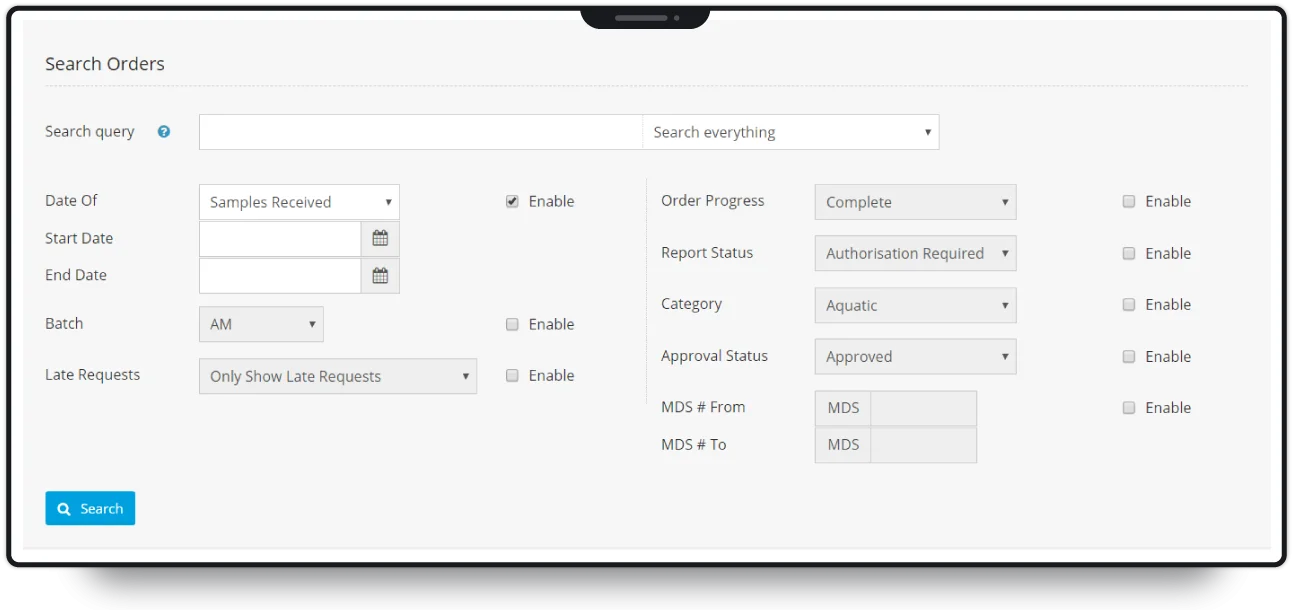
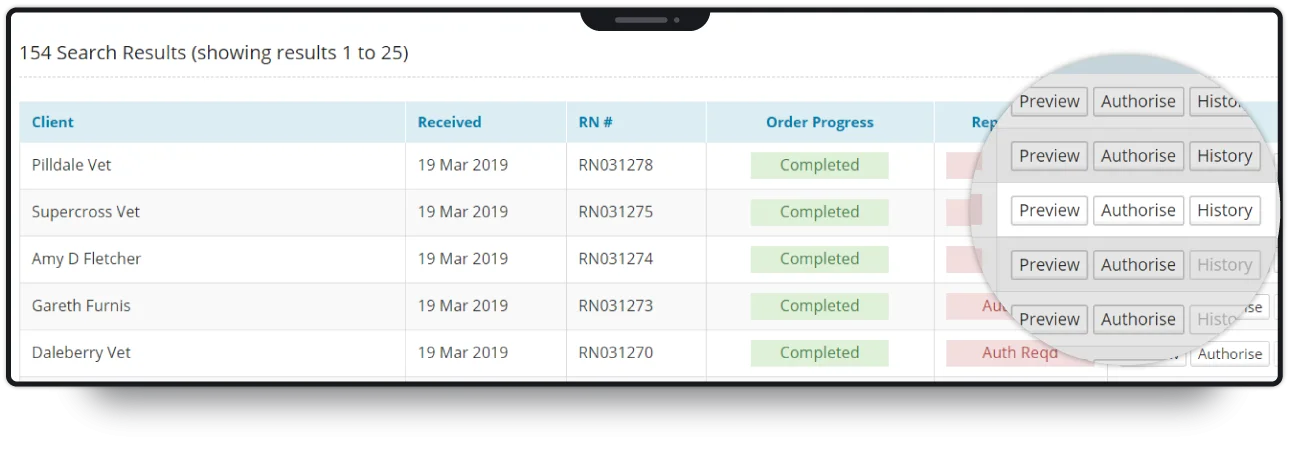
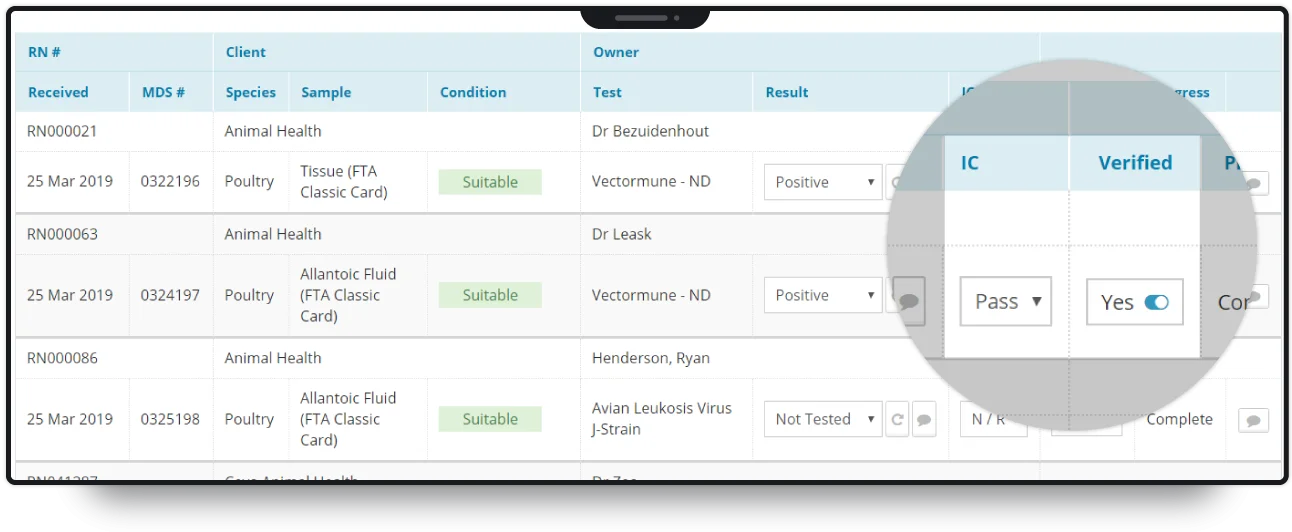
Upon arrival at the laboratory, samples are batch processed and sent for testing. Our software tracks samples at each stage, automatically informing clients of the progress of their order. As an ISO-accredited laboratory, MDS is required to perform quality control (QC) on samples, and audit any irregularities.
Our software presents full audit control functionality to the laboratory, allowing for change-tracking throughout the process of testing a sample.
As a system which is required to be globally accessible and operational 24/7, all changes to the software undergo rigorous testing to ensure stability and to prevent unnecessary downtime. In addition, in dealing with medical records, our software employs strict security measures, with stringent access controls to prevent unauthorised access to data.
Key Innovations